Cookie preferences
This website uses cookies, which are necessary for the technical operation of the website and are always set. Other cookies, which increase the comfort when using this website, are used for direct advertising or to facilitate interaction with other websites and social networks, are only set with your consent.
Configuration
Technically required
These cookies are necessary for the basic functions of the shop.
"Allow all cookies" cookie
"Decline all cookies" cookie
CSRF token
Cookie preferences
Currency change
Customer-specific caching
FACT-Finder tracking
Individual prices
Selected shop
Session
Comfort functions
These cookies are used to make the shopping experience even more appealing, for example for the recognition of the visitor.
Note
Show the facebook fanpage in the right blod sidebar
Statistics & Tracking
Affiliate program
Conversion and usertracking via Google Tag Manager
Track device being used
| Item number | Size | Datasheet | Manual | SDS | Delivery time | Quantity | Price |
|---|---|---|---|---|---|---|---|
| A300-589A-T | 10 µl (2 µg) | - |
2 - 8 business days* |
164.00€
|
|||
| A300-589A | 100 µl | - |
2 - 8 business days* |
588.00€
|
If you have any questions, please use our Contact Form.
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
You can also order by e-mail: info@biomol.com
Larger quantity required? Request bulk
Protein function: Necessary for the fragmentation of Golgi stacks during mitosis and for their... more
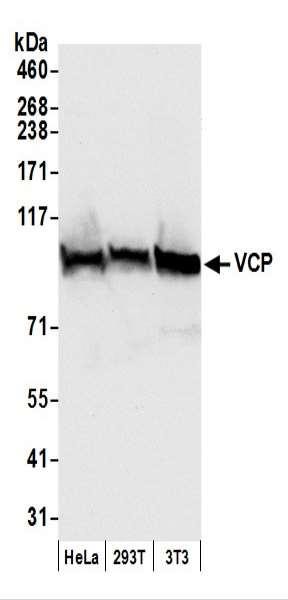
Product information "Anti-VCP"
Protein function: Necessary for the fragmentation of Golgi stacks during mitosis and for their reassembly after mitosis. Involved in the formation of the transitional endoplasmic reticulum (tER). The transfer of membranes from the endoplasmic reticulum to the Golgi apparatus occurs via 50-70 nm transition vesicles which derive from part-rough, part-smooth transitional elements of the endoplasmic reticulum (tER). Vesicle budding from the tER is an ATP-dependent process. The ternary complex containing UFD1L, VCP and NPLOC4 binds ubiquitinated proteins and is necessary for the export of misfolded proteins from the ER to the cytoplasm, where they are degraded by the proteasome. The NPLOC4-UFD1L-VCP complex regulates spindle disassembly at the end of mitosis and is necessary for the formation of a closed nuclear envelope. Regulates E3 ubiquitin-protein ligase activity of RNF19A. Component of the VCP/p97-AMFR/gp78 complex that participates in the final step of the sterol-mediated ubiquitination and endoplasmic reticulum-associated degradation (ERAD) of HMGCR. Also involved in DNA damage response: recruited to double-strand breaks (DSBs) sites in a RNF8- and RNF168-dependent manner and promotes the recruitment of TP53BP1 at DNA damage sites. Recruited to stalled replication forks by SPRTN: may act by mediating extraction of DNA polymerase eta (POLH) to prevent excessive translesion DNA synthesis and limit the incidence of mutations induced by DNA damage. Required for cytoplasmic retrotranslocation of stressed/damaged mitochondrial outer-membrane proteins and their subsequent proteasomal degradation. Essential for the maturation of ubiquitin-containing autophagosomes and the clearance of ubiquitinated protein by autophagy (PubMed:20104022). [The UniProt Consortium]
| Keywords: | Anti-VCP, Anti-TER ATPase, EC=3.6.4.6, Anti-Valosin-containing protein, Anti-15S Mg(2+)-ATPase p97 subunit, Anti-Transitional endoplasmic reticulum ATPase |
| Supplier: | Bethyl Laboratories |
| Supplier-Nr: | A300-589A |
Properties
| Application: | WB, IP |
| Antibody Type: | Polyclonal |
| Conjugate: | No |
| Host: | Rabbit |
| Species reactivity: | human, mouse (Expected: rat, bovine, pig) |
| Immunogen: | synthetic peptide. The epitope recognized by A300-589A maps to a region between residue 750 and the C-terminus (residue 806) of human Valosin-Containing Protein using the numbering given in entry NP_009057.1 (GeneID 7415). |
| Format: | Antigen Affinity Purified |
Database Information
| KEGG ID : | K13525 | Matching products |
| UniProt ID : | P55072 | Matching products |
| Gene ID | GeneID 7415 | Matching products |
Handling & Safety
| Storage: | +4°C |
| Shipping: | +4°C (International: +4°C) |
Caution
Our products are for laboratory research use only: Not for administration to humans!
Our products are for laboratory research use only: Not for administration to humans!
Information about the product reference will follow.
more
You will get a certificate here
Viewed